digitAAL Life -
Einzigartiges Eye-Tracking als Biomarker für Monitoring und Diagnostik in der Demenztherapie
Facts of the crowdinvesting campaign:
Target investment amount: € 180.000 to 525.000
Offered participation: 5,66 % to 14,89 %
Pre-money valuation: € 3.000.000
Minimal investment and denomination: € 1.000
End of the subscription period: 12.08.2021
180.000 €
Funding threshold
525.000 €
Funding goal
418.000 €
247 investors
232,22 %
financed
418.000 €
invested
successfully financed
status
Information
About digitAAL Life
digitAAL Life GmbH is a spin-off from JOANNEUM RESEARCH and specializes in the development of innovative digital solutions for healthcare. Together with strategic partners from research, medicine and practice, digitAAL Life has developed a tablet-based ‘serious game’ for multimodal activation of cognitive performance, which has already been launched on the German-speaking market.
For the upcoming seed phase, we are aiming to certify our product as a medical device for dementia therapy and to develop our dementia diagnostics tool on the basis of eye tracking as a biomarker.
In addition to the national seed financing of € 600,000 which we have already raised, we are now raising equity as a co-investment to that funding.

Mit dem Laden des Videos akzeptieren Sie die Datenschutzerklärung von YouTube.
Mehr erfahren
Problem
Dementia is a global challenge with currently about 50 million people affected worldwide, with no cure in sight, and that number increasing due to the aging population (expected to reach 131 million by 2050). Therefore, the WHO and “The Lancet Commissions” recommend combined and personalized physical, mental and social (so called “multimodal”) interventions as the only effective approach.

Solution
The innovation of digitAAL Life GmbH is a tablet-based serious game as a therapy, that is aimed at slowing down the onset and progression of dementia. For simultaneous diagnostics, our novel eye tracking solution is used as a biomarker, in combination with other outcome parameters to monitor dementia status.
The technological innovation is the measurement of human factors by means of eye tracking, already investigated by JOANNEUM RESEARCH. This allows the first indications of objectively measurable indicators for the status of dementia, based on gaze behavior and movement patterns (eye tracking as a biomarker), which to our knowledge is unique worldwide. You can see this video which elaborates on the subject.
For the innovation leap, digitAAL Life is striving to advance the development of AI-based decision support for both the continuous and punctual status monitoring of dementia, as well as carry out the certification process as a medical device.
For the future version of the product, the measurement of the human factors and the client performance data during the completion of the serious game will form the basis of the decision support for monitoring dementia status. In contrast to the current status assessments for dementia every 6 months by means of neuropsychological testing, the Serious Game with Decision Support is designed to enable continuous monitoring of dementia status and immediate reaction to sudden status changes. In the event of a more sudden deterioration in status, for example, a request can be made to contact medical care and/or to seek neuropsychological testing again immediately – rather than at the specified time in a few months.
To our knowledge, digitAAL Life is a novel solution on the market that offers both personalized, multimodal training as therapy, as well as tools for the diagnosis of dementia in a single, integrated solution. It can also be used for prophylaxis and other neurodegenerative diseases (such as Parkinson’s disease). The product is offered as Software-as-a-Service (SaaS), customers receive licenses for the app and access to the backend. No hardware is sold, but complementary training is offered in person and online.

Business Modell
The product is sold directly to B2B customers, who are addressed personally by the sales team. Currently, this is done via the personal networks of Maria Fellner, Robert Hartmann and Elke Zweytik. With the investment, 1-2 additional sales people will be hired, who will also have access to the personal network of the team members in the field of care and dementia.
Once the app has been downloaded, content activation takes place after payment of the license fees to digitAAL Life.
Various sales partners can act as intermediaries, who are offered commissions which are individually agreed. Therefore, we believe that non-profit organizations (network partners) that use the product themselves might prefer sales-dependent reductions on their own license prices, whereas commercial trade/distribution partners are likely to prefer a direct settlement of commission. Various training centers for activity in old age and dementia are also important promoters of our product.
We are currently setting up an online store for the registration and ordering of training courses for private individuals (individually or in group courses), including a payment function. This is being set up as part of the seed project.
In sales for B2B customers, the implementation of discounted, time-limited pilot projects in organizations, where the product can be tested and tried out in practical use, has proven its worth based on many years of experience. Afterwards, a decision is made regarding the adoption of the product for regular operation, which is then also used to recommend the product to private customers.
The product is sold as software as a service (SaaS), which means that the use of the app and content is subject to annual license payments. In the company’s revenue calculations, it is assumed that the follow-up rate is 80% for B2B customers, but only 50% for private customers. This is due to the fact that in the case of progressive dementia, use of the product is only useful and possible for a certain period of time. An average period of private use is 3 years.
Market
The World Alzheimer Report 2015 showed about 46.8 million people with dementia worldwide; this number is expected to double about every 20 years. About two thirds of all these cases concern the form of Alzheimer’s dementia.
According to current estimates, 130,000 people in Austria are living with some form of dementia (Höfler et al. 2015). Due to the continuous age increase in the population, this proportion is forecast to double by 2050 (Sütterlin et al. 2011).
Annual new cases of dementia in Austria are expected to increase from 35,700 in 2020 to 65,500 in 2050. Prevalence rates rise steeply with age. The incidence doubles every five years or so, rising from just over 1 percent in the 65-69 age group to around 40 percent among the over-90s.
Two-thirds of all sufferers have already reached the age of 80 and two-thirds of sufferers are women. Given the current age structure, one in ten people over the age of 65 is affected by dementia.
The annual cost of dementia in Austria is €2.9 billion. The cost of home care is estimated at €10,000 per person per year, while this rises significantly to between €25,000 and €43,000 for inpatient settings.
Germany had 1.5 million people living with dementia in 2013, and the estimate for 2050 is 3 million people. In the Netherlands, the numbers are comparable to Austria.
The overall market includes all people with mild to moderate Alzheimer’s dementia as well as mild cognitive impairment. However, the product can also be usefully used for prevention for all people aged 65 and over, which represents an even larger market.
Currently, our target customers are primarily organizations in health and care that offer inpatient or mobile care (B2B market); geographically, the German speaking, BENELUX and CEE markets are being worked on in the seed phase. These organizations are simultaneously positioned to promote our solution to private customers, they can train them in their region and offer first level support (since cooperation agreements are already in preparation).
Since this is a new market to be developed in the sense of an emerging market, it is not possible to define targeted market shares. However, it has been possible to start sales activities in Austria during the pre-seed phase and to hold talks with the major care providers. In addition to people with dementia, the focus has also shifted to 24-hour care, which has a need for engagement opportunities and where the quality can also be improved. A typical large provider of 24-hour care has several hundred clients and twice as many personal care workers.
Once the product is in regular use at large organizations in at least one area, we anticipate the possibility for accelerated growth due to other areas of the organization becoming interested in the product. At the same time, affected persons and relatives may become interested in purchasing the product privately.
By the end of 2022, we are aiming to reach an installation base of just under 2,900 licenses in the German speaking region, BENELUX and CEE region (where approximately 1.8 million people suffer from dementia), and by the end of 2024, our goal will be to reach an installation base of approximately 29,000 licenses in Europe (where around 10 million people suffer from dementia).

Traction
Story behind the company
Since 2011, Maria Fellner (CEO and founder of digitAAL Life) has been involved in Active and Assisted Living at JOANNEUM RESEARCH (an Austrian-based business oriented provider of innovation and technology that specializes in various fields of science), as well as mobile dementia (M.A.S) trainings at Sozialverein Deutschlandsberg (a regional provider of social services for the 15 communities in the Deutschlandsberg district).
In 2015, she started a successful cooperation between these two organizations and subsequently implemented a digital, tablet-based solution for multimodal training as result of joint research projects, which yielded convincing field tests and pilot projects.
Her research at JOANNEUM RESEARCH during this time focused on investigating ICT-based indicators for dementia.
Foundation of the company
digitAAL Life was founded as a spin-off in February 2020 as a result of successful research projects at JOANNEUM RESEARCH with partners from medicine and practice. Our multidisciplinary and experienced team from technology, dementia and care launched the first version in summer 2020, which is already in use at the Austrian Red Cross.
The first paying customers (Austrian Red Cross, Caritas, private individuals) have already been acquired for the current version of the product. For further product development in the medium term, digitAAL Life is also integrated into the R&D portfolio of JOANNEUM RESEARCH and Medical University Graz.
In the upcoming seed phase, we intend to focus on the development of high-tech features for dementia therapy and diagnostics and certification as a medical device for dementia therapy. In addition, sales will be established with a focus on the German speaking, BENELUX and CEE regions.
Testimonials
- Petra Schmidt, Austrian Red Cross: “Our volunteer visiting service has been working with the digitAAL Life app for years. Whether in an individual setting or in group training, our colleagues appreciate the many ways the app provides support. And the fun factor should not be underestimated either.”
- Univ.-Prof. Dr. Reinhold Schmidt, Medical University Graz, Clinical Department of Neurogeriatrics: “Multimodal training programs are a beacon of hope in Alzheimer’s therapy. digitAAL Life’s tablet-based program is pioneering the evaluation of the effectiveness of complex training models to improve cognitive performance in Alzheimer’s patients.”
- Sen. Scient. Dr. Sandra Schüssler, MSc, BSc, Medical University Graz, Institute of Nursing Science: “New technologies are playing a big role in healthcare. Not only to improve cognitive performance, but also to alleviate the lack of resources. The digitAAL Life app provides family members and caregivers the ideal tool for their work.”
- Monika and Friedrich Mulzet: „My wife has been suffering from Alzheimer‘s dementia for four years. Her cognitive performance has become worse and worse. By chance, I found out about the DIGITAL life app and started training with her regularly. Although I was skeptical at first, I am now surprised by my wife‘s progress. In the meantime, I even practice with the app myself to stay mentally fit.“
COVID-19 Impact on Business
The COVID-19 pandemic had temporarily delayed our sales activities after our market entry in July 2020 because our primary B2B customers, namely organizations in health care and social sector, have been heavily impacted by the pandemic (visiting restrictions, testing, vaccination). Furthermore, trade fairs and professional conferences have been cancelled or transformed into an online format. On the other hand, this crisis motivated the same organizations to use digital technologies for their clients and patients (e.g. tablets with video conferencing for keeping in touch with relatives), but also for themselves (video conferencing with external professionals who are not allowed to visit the organization). From a mid-term perspective, we have observed a boost for digital technologies in the care and social industries, where the benefits of digital tools in the current economy are self-explanatory.
From a product feature perspective, we have been inspired by the crisis to include remote care features into our product development roadmap, such as remote training as an addition to training in physical presence and integrated video conferencing.
On the strategic level, we have discovered a new field of application for our solution, such as treatment and diagnostics of long-COVID. We have already submitted proposals for public funding together with our strategic research partners, where we are now waiting for the jury decisions. These research projects would give us the possibility to keep our strategic position as an innovation leader and to diversify our product portfolio on basis of the same technology and software platform.
Grants
digitAAL Life has recently received approval for the research proposal “MARA – Mobile AI-Enabled Recommender for Cognitive Activation and Stress Management“, where we have planned a budget of €390,000 with 60% funding rate (i.e. €234,000 of this is a non-repayable grant), for 24 months project duration, starting in July 2021. In this project we will be working with Austrian and Dutch partners in the field of dementia, which we expect to support and complement our other development and market entry activities in this area.
In addition, we are in the process of joining the already running Austrian research project multimodal (see www.multmodAAL.at) with Medical University Graz, JOANNEUM RESEARCH and the Austrian Red Cross. The results of this clinical RCT study with our tablet-based intervention will be an important part of the medical device certification as well as an important source of real-world data for the future decision support module with eye tracking. Besides these factors, we are planning to receive a budget of €120,000 with 60% funding for digitAAL Life (i.e. €72,000 non-repayable grant).
Additional research proposals have been already submitted in the first four months of 2021 (cf. section on COVID-19 impact above), but we are still waiting for juries´ decisions.
Historical Financials
The chart below shows the 2020 financials that have been sourced from the company’s annual report. Please consider that past performance is not a reliable indicator or guarantee of future results.

Team
The founding team Maria Fellner, MBA, Dr. Heinz Mayer and Josef Steiner, founded digitAAL Life GmbH in February 2020.
Shareholders
- Dr. Heinz Mayer is director of the institute DIGITAL of JOANNEUM RESEARCH (www.joanneum.at/digital) and responsible for the R&D portfolio and the strategic focus of the institute.
- Josef Steiner is a retired teacher, who served as chairman of the Social Welfare Association Deutschlandsberg from 1998-2020, was spokesman of the ARGE of social welfare associations in Styria from 2009-2020, and chairman of the social welfare association Deutschlandsberg from 2010-2020. He has an outstanding network in the Austrian health and care system.
- Maria Fellner, MBA is managing director and co-founder of digitAAL Life GmbH. From 2013 to 2020, she was head of business development at the institute DIGITAL of JOANNEUM RESEARCH (Graz, Austria). She supported the research groups of the institute in the areas of business development, marketing, PR and sales of their innovative (future) products and services and designs customer relationship management. Since 2011, she has also coordinated the institute’s activities in the area of Active and Assisted Living (AAL) and Digital Care. From 1999 to 2013, she led the Intelligent Acoustic Solutions team at the institute and contributed with her acquisitions and project management to the success of this growing field of activity, which with its many industry-related projects is a good example of the active implementation of business and market orientation in applied research. This professional work is based on a diploma in Electrical Engineering (Graz University of Technology) and a Professional MBA in Entrepreneurship & Innovation (WU Executive Academy, Vienna). In addition to this full-time job, she is a lecturer for innovation and technology in healthcare at IMC FH Krems (Austria) in the Master programs “Applied Health Sciences” and “Management of Healthcare Facilities”. She is vice president of “AAL AUSTRIA – Innovation Platform for Intelligent Assistance in Everyday Life” (www.aal.at).
Core Team
- DGKP Elke Zweytik is a trained M.A.S. (“Motivate, Activate, Strengthen”) trainer and has many years of experience in a wide variety of areas of inpatient and mobile care. Her industry and professional knowledge is an important driving force for the further development of the product’s content, with a constant focus on practical relevance. Having worked for the Sozialverein Deutschlandsberg for several years, she knows the work as an M.A.S. trainer with clients from her own experience and is familiar with their needs, abilities and deficits in practice. From her experience paired with great empathy she draws her creativity, which leads to many new ideas for future product features, but also digital and analog supplementary offers for our digital solution.
- Mag. Robert Hartmann brings strategic knowledge in social policy and a large and high-ranking network throughout Austria, which has already enabled numerous customer meetings with welfare organizations at top management level during the pre-seed project, which, for example, also led to the pilot project at Caritas Vienna. Through his contacts, we are ourselves in talks with the Ministry of Social Affairs to initiate a pilot with volunteers and several welfare organizations in several Austrian regions. This is based on his long professional experience, including as managing director of Hilfswerk Upper Austria, and his many years of activity in relevant networks, such as the Federal Working Group of Independent Welfare Organizations in Austria.
- Thomas Fruhmann worked for 19 years in different areas of JOANNEUM RESEARCH as an IT administrator and now uses this acquired knowledge for the structured setup of the IT for the company processes and the operational use of the product. He ensures the fail-safe operation of the entire IT infrastructure and data protection, supports the sales of the product, e.g. with license management, and is also already looking into the future solution options for the web store in connection with the company website and the necessary payment service provider. Ideally, the CRM system would also be integrated here with order management and invoicing, so that the sales processes are completely mapped in a digital workflow.
- Raphael Marton, BSc will soon complete his master’s degree in electrical engineering-biomedical engineering at Graz University of Technology and is responsible for the roadmap to medical device certification. As part of his master’s thesis, he will focus on risk management for the future medical device, which is an important milestone for certification.
- Petra Hauptmann is dementia trainer and trained nurse.
- Ulli Pratter´s responsibilities are graphics design and marketing.
After successful funding by Funderbeam/Danube Angels, we intend to extend the team by hiring a CTO, two software developers (frontend/app and backend/database development) and 1-2 employees for sales.
Of the external cooperation partners for R&D, special mention should be made of Univ-Prof. Reinhold Schmidt (Medical University Graz, Clinical Department of Neurogeriatrics) and Dr. Sandra Schuessler (Medical University Graz, institute of Nursing Science), but also Dr. Lucas Paletta and Silvia Russegger, MSc from JOANNEUM RESEARCH – institute DIGITAL.

Competition
Our competition can be divided into two main groups: Dementia training conducted by qualified trainers or occupational/physiotherapists as well as psychologists in the conventional way “with paper and pencil” or various “analog” materials; and digital applications for cognition training, monitoring or diagnostics.
The intended use of our digital product is supervised training conducted by trainers, as the tablet app supports them in their work and increases the efficiency and quality of the training. However, it still remains an alternative, and especially in the public sector we know that it takes time to introduce new digital solutions.
Our main competition comes from other digital solutions for cognitive training. In our competitive analysis, we took a comprehensive look at competing solutions, from simple cognitive games and memory training apps to personal training apps, home care apps, and diagnostic apps. In our initial search, we did not limit ourselves to apps for dementia patients, as they could use timeless care, for example, especially in the early stages of dementia. However, general apps for telemedicine and telecare (e.g. evocare.de) are not really our competitors, as they can be used in addition to our app.
We then narrowed our application search to apps targeting dementia. As the number of dementia patients grows rapidly, new apps and tools are entering the market. We categorized the apps into two main groups: (i) apps that provide activity training that can be done alone or with caregivers, and (ii) apps that provide diagnostic features. Both types of apps are often offered for home use as well as for professionals, but in different versions.
For the feature comparison, we defined three main functionalities: 1) multimodal training, i.e., memory and cognition exercises combined with movement exercises, 2) functional aids for patient and caregiver daily life, and 3) diagnostics. We then specified the features covering these functionalities and used them in our competitive analysis. To our knowledge, digitAAL Life offers the only solution that provides multimodal training with personalized support, personalization of content, and most importantly, decision support for diagnostics and continuous monitoring.
In general, it is not possible to compare the pricing of the products and services because many of the products, like ours, are in a start-up phase. Pricing also depends on the intended use, e.g., by professionals or private individuals.
Other early dementia/Alzheimer’s solutions include those from start-up companies such as Altoida (CH/US), Braincheck (Texas, US), Mediaire (Germany), Neotiv (Germany), Eyewise-diganostics (Israel), and ClearskyMD (UK). Since they do not include activating training, they are not our direct competitors.
Overall, the competitive landscape consists of startups. This suggests that there is a need in the market, but larger companies have not yet developed their own solutions. It is therefore possible that they will follow the same strategy as larger companies for other new innovations: They will watch the startups and then adopt the best ones once the technological benefits are validated.
digitAAL Life also offers a premium solution to the market that takes a novel approach to servicing patients, families, physicians and care organizations, and the society. Unlike free or low-cost memory training apps, digitAAL Life GmbH positions itself as a leader in innovation and quality, based on years of scientific expertise and offering a breakthrough solution with unique benefits. Upon completion of this seed phase, digitAAL Life GmbH will be the only provider to hold certification as a medical device.
Capital
Previous financing
In 2020, the company raised EUR 200,000 pre-seed investment from the Austrian Promotional Bank. An additional EUR 50,000 has been invested from the three founders to build the company and cover mainly personnel costs.
Current financing
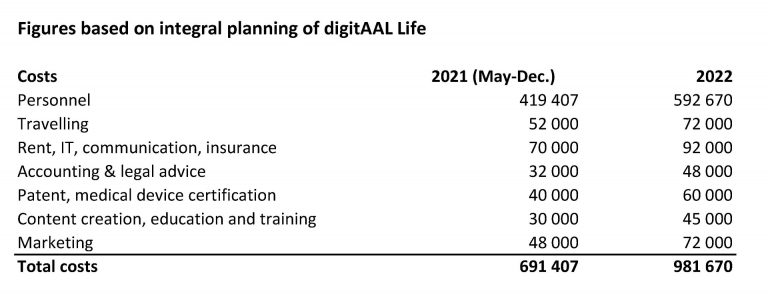
The total capital requirement for the seed period (24 months) is approximately €2.2m and will come from a financing mix of national seed financing, this funding round on Funderbeam/Danube Angels as well as licensing revenues, training offers and research funding.
digitAAL Life has recently been awarded €600,000 equity free seed financing from the Austrian Promotional Bank, which is part of the current seed round.
Funding allocation plan
The allocated funds will be used for:
- Personnel costs: additional staff for software development (2 persons), a CTO and 1-2 persons sales to be hired, current staff (7 persons), this will be also co-financed by additional research funds, such as the Eurostars project MARA.
- Marketing and sales activities in DACH, BENELUX and CEE region: trade fairs, exhibitions at industry specific conferences, advertisements in magazines and journals, customer visits, travelling costs, patenting and trademark protection etc.
- Own activities and consulting costs for medical device certification, such as QMS certification, risk management etc.
- Running costs for office space, IT, communication, legal and financial consultants.
Exit
digitAAL Life is at a very early stage where it is difficult to predict the likelihood of different exit scenarios. Our goal is to grow the company to the point of self-sustainability and to increase the value of our stakeholders’ assets in the company. We also believe that one day digitAAL Life may become an attractive acquisition target by larger medical/pharma companies. In the meantime, digitaal Life shares that are issued to Funderbeam investors will be tradable on the Funderbeam Marketplace.
Risk
- Risks associated with competition and entry to new markets: One of the risks is that other companies aim develop a similar tablet-based solution. Although we are confident that our approach has a competitive advantage, we are mindful of the need to launch sooner to secure the first-mover advantage. We aim to enter new markets with diverse languages and cultures by partnering with local stakeholders in the healthcare field. There is the risk, that attracting such partners might take longer than planned and that language and cultural adaptions to our solution might be more expansive than expected. There is also a risk that larger companies who wish to enter the same market might have more resources and a larger network than us, which could result in them beating us to the market.
- Risks associated with need for future financing: We anticipate the need for future equity financing, if we are unable to secure this, we may fall behind our competitors. We believe that this would not lead to us shutting down the business, but it would likely impact our long-term position in the market.
- Risk associated the medical device certification: There is also a risk, that our product will not be licensed as medical device for therapy and diagnostics of dementia. This could hinder us to from entering the market, thus limiting our growth.
- Risk associated with partner and user adoption: Our business success depends on how quickly we are able to onboard partners and users. For this, it is necessary that we convince as many partners as possible to use our solution with their customers. Our partners have the potential to act as multipliers for selling licenses to end-users. We believe this is a relatively low risk, as we are already working with the Austrian Red Cross and the University Regional Hospital in Graz who are already using our solution. Nevertheless, if we are unable to grow our partner network, this could have an adverse impact on our growth.
- Risks associated with marketing: Daily marketing is required in order for us to build and maintain the brand presence and continue spreading awareness to existing and potential new customers. As we are introducing a novel approach, we believe that a significant amount of funds needs to be allocated for aggressive promotion to the right customer segments. If we are unable to identify the correct market segments or this doesn’t result in the expected onboarding of new partners and customers, we may burn through the allocated funds before achieving the desired growth results.
- Risk associated with staff and recruitment: digitAAL Life relies on its team members for the delivery of business success, and as a small business may suffer adverse effects if it cannot attract new hires into critical roles, or if key staff members leave the business.
- Risk associated with grants: digitAAL Life has been granted a conditionally repayable grant, which is paid according to a milestone plan. There is a risk, that these milestones are not reached in the agreed time and the tranches of the grant is not paid out.
- Risks associated with the economic situation: 2020 has proven to be a challenging year for the global economy due to the emergence of Covid 19. It is difficult to predict the impact that an unstable economy might have over an extended period of time. Therefore, it is possible that such a scenario may therefore have an impact on our business in some shape or form.
Details der Crowdinvesting-Kampagne
Facts of the crowdinvesting campaign:
Target investment amount: € 180.000 to 525.000
Offered participation: 5,66 % to 14,89 %
Pre-money valuation: € 3.000.000
Minimal investment and denomination: € 1.000
End of the subscription period: 12.08.2021
180.000 €
Funding threshold
525.000 €
Funding goal
418.000 €
247 investors
232,22 %
financed
418.000 €
invested
successfully financed
status
How can I invest?
This transaction is carried out together with our partner platform. Therefore, there are two ways for you to invest in digitAAL Life, either via Danube Angels or via Funderbeam.
Questions concerning our projects? Do you have any questions concerning our participation model or the execution of the investment?
Answers to frequently asked questions from our investors can be found here.
We will also be glad to answer your questions personally in a call or by e-mail.

